L’effet post-bombe atomique a profondément renouvelé notre lecture du carbone 14. Le pic de radiocarbone issu des plus de 500 essais nucléaires réalisés entre des années 1950 à 1960 a laissé une signature temporelle unique. Celle-ci est aujourd’hui exploitée par les scientifiques pour la datation carbone 14 pour le marché de l’art et l’analyse des matériaux organiques tels que les bois, ivoire, os, dents, lin, paille, papier ou autres textiles.
Pour les professionnels de l’art dépendant de techniques d’authentification d’œuvres pour identifier des objets pouvant être composites ou restaurés, l’effet post-bombe représente une opportunité de fiabiliser la provenance de ceux-ci. La maîtrise des référentiels post-bombe (hémisphère Nord/Sud) et des corrections isotopiques est ainsi essentielle afin de garantir la fiabilité des résultats et leur interprétation (provenance, cohérence historique, due diligence).
Dans cet article, nous clarifions les mécanismes et implications analytiques de l’effet post-bombe, détaillons la mesure carbone 14 par AMS et les référentiels de calibration appliqués à l’art. Vous y trouverez également des cas d’usage (toiles en lin, châssis en bois, papiers, ivoires, os/dents) et des explications concernant le rôle d’un laboratoire expert comme CIRAM pour transformer une mesure carbone 14 en preuve scientifique utile aux experts, maisons de ventes, galeries, musées et collectionneurs.
Carbone 14 et effet post-bombe : mécanismes & implications analytiques
Maîtriser le comportement isotopique du carbone 14 permet de poser les bases d’une datation rigoureuse et d’une authentification crédible.
Datation carbone 14 en contexte « art » : principes, effet Suess et enjeux analytiques
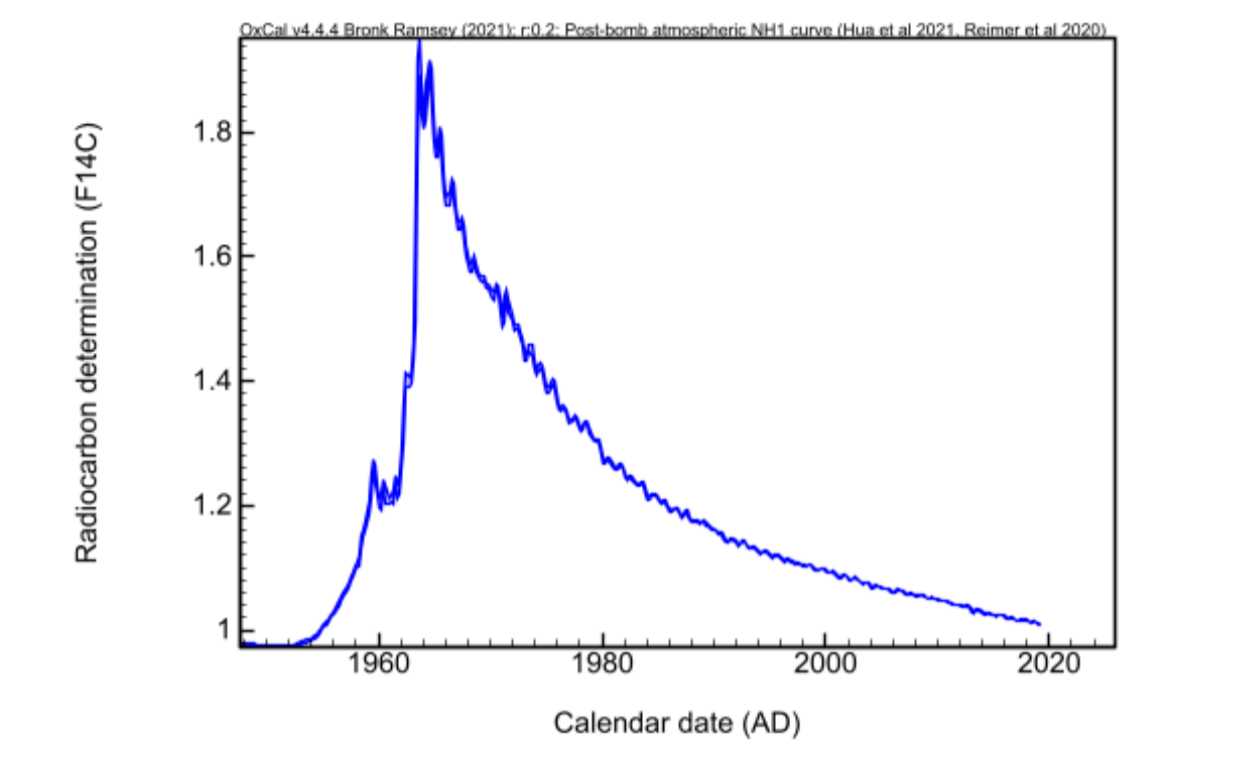
L’augmentation soudaine du carbone 14 atmosphérique suite aux essais nucléaires a généré une courbe de radiocarbone très distincte, connue sous le nom de « bomb-peak ». Les bombes nucléaires ont ainsi entraîné une production massive de radiocarbone à partir de 1954 et jusqu’à pratiquement doubler sa concentration en 1963 et 1964. Ceci permet de placer une limite stricte entre les organismes ayant vécu avant et après 1954. Cette anomalie isotopique, longtemps perçue comme une perturbation, est devenue un repère temporel d’une précision inédite pour les scientifiques.
Le carbone 14 produit par les rayons cosmiques, s’intègre en effet à la matière organique vivante (plantes, animaux). Le mesurer permet ainsi de dater des supports variés : bois, fibres végétales (lin, paille, papier), ivoire, os et dents.
Deux phénomènes notables bousculent l’équilibre naturel :
- Effet Suess : la combustion de CO₂ fossile (sans carbone 14) diminue la proportion de carbone 14 atmosphérique
- Effet post-bombe : à l’inverse, les essais nucléaires augmentent fortement la concentration de carbone 14 (avec un pic entre 1963-1964).
Contrairement à la datation classique du carbone 14 (qui est limitée à quelques centaines d’années de précision pour les périodes récentes), le signal post-bombe permet de dater un matériau organique à ±1-2 ans près entre 1954 et 2020. La concentration anormale du carbone 14 dans les êtres vivants ayant vécu après 1954 est idéale pour repérer les faux récents.
L’identification de cette sur-concentration est particulièrement utile pour :
- Déterminer si un objet est antérieur ou postérieur à 1954 : un seuil critique pour les objets dits « modernes » ou « d’après-guerre »
- Vérifier si une œuvre revendiquée comme étant de 1963, par exemple, a bien été produite pendant le pic de radiocarbone (ce qui est mesurable)
- Distinguer un ivoire ancien (pré-Convention CITES) d’un ivoire moderne prélevé illégalement.
Quels matériaux peuvent être datés via l’effet post-bombe ?
L’effet post-bombe concerne exclusivement les matériaux d’origine organique ayant intégré du carbone atmosphérique après 1954. Voici les principaux matériaux analysables avec l’analyse post-bombe :
- Bois (supports, châssis, sculptures) : permet de distinguer un bois de croissance moderne d’un bois ancien, d’identifier un châssis remplacé ou une restauration datable ou encore de repérer un bois jeune dans une œuvre prétendument ancienne.
- Toiles de lin, papiers, textiles : il est possible de dater la fibre (et non la peinture), de révéler une toile post-1960 derrière une attribution du 19e ou par exemple, de vérifier l’homogénéité des éléments constituants (toile, doublage, passe-partout, collage).
- Ivoires, os, dents : l’analyse permet de montrer qu’un ivoire prétendu ancien est en réalité moderne (signal post-bombe), ou au contraire de confirmer l’absence de bomb-pulse.
Cependant il existe des limites et précautions à noter :
- La datation carbone 14 cible la matière échantillonnée : dater la toile ≠ dater la peinture et dater le bois ≠ dater la sculpture.
- Les restaurations (colles PVA, vernis, consolidants) peuvent contaminer les matières organiques d’où la nécessité d’un prélèvement ciblé et d’un pré-prétraitement adapté (extraction alpha-cellulose pour fibres, purification du collagène pour os/dentine ou procédures acid-alkali-acid, ultrafiltration, etc.).
- L’ancien bois réemployé existe (ce qui est rare mais possible) : il est alors crucial de croiser cette méthode avec la xylologie/dendrochronologie, l’imagerie et l’étude de provenance.
Chaque matériau, selon son origine, sa préparation et ses éventuels traitements, nécessite une stratégie d’échantillonnage rigoureuse, que CIRAM établit en lien avec conservateurs, restaurateurs et experts.
Méthodes de mesure carbone 14 post-bombe pour le marché de l’art
Méthode AMS (Spectrométrie de Masse par Accélérateur) : précision, fiabilité et cas d’usage
La spectrométrie de masse couplée à un accélérateur (AMS) est la méthode de référence pour l’après-bombe en art. Elle mesure directement les rapports carbone ¹⁴C, ¹³C et ¹²C avec une excellente sensibilité à partir de micro-quantités (prélèvements discrets, non visibles après intervention).
Cette technologie permet :
- Micro-échantillonnage : analyse de très faibles quantités de matière (quelques milligrammes) : fil de lin, paillette de bois, micro-éclat d’ivoire, fibre de papier.
- Capacité à traiter des matériaux restaurés (avec protocoles de décontamination adaptés).
- De restituer des résultats en pMC (percent Modern Carbon), accompagnés d’un intervalle calendaire précis.
Le processus se déroule en plusieurs étapes :
- Prélèvement ciblé (zone masquée, bord, excédent filaire)
- Prétraitement : solvants doux/forts selon matrices, extraction alpha-cellulose (fibres/papier), purification collagène (os/ivoire), élimination des vernis/colles.
- Graphitisation, permettant d’isoler le carbone pur
- Mesure AMS avec correction isotopique via δ¹³C
- Interprétation selon le référentiel post-bombe (Nord ou Sud) et rendu d’un rapport scientifique documenté.
Référentiels, calibration et interprétation : faire parler la matière
CIRAM utilise les courbes de calibration post-bombe publiées par la communauté scientifique (comme NH1, NH2, NH3, SH1-2, SH3) afin de convertir les pMC mesurés en dates calibrées. Cette lecture permet de répondre à des questions précises :
- Une œuvre a-t-elle été réalisée avant ou après 1954 ?
- Son matériau correspond-il à une production postérieure à la mort de l’artiste ?
- Est-il possible de distinguer une pièce originale d’une reproduction moderne, basée sur le même matériau ?
Pour convertir une mesure en preuve, les laboratoires spécialisés en datation carbone 14 pour l’art :
- Utilisent des courbes de calibration post-bombe (différentes NH/SH) reflétant l’évolution du carbone 14 atmosphérique depuis 1954
- Appliquent des standards internationaux et délivrent des résultats comparables (pMC/Fcarbone 14, intervalle probable d’année)
- Interprètent les résultats au cas par cas : ils mettent en relation support, contexte, origine géographique présumée, historique de restauration et stratégie d’échantillonnage.
Grâce à l’effet post-bombe, chaque variation de pMC devient une horloge, et chaque œuvre, un témoin de son époque. CIRAM suit des protocoles QA/QC complets (blancs, étalons bomb-peak, duplicatas, validation de rendement) et consigne chaque étape. Les rapports sont citables, rédigés dans un langage opérationnel (utile aux catalogues de vente, rapports d’experts, comités et assureurs).
Effet post-bombe et marché de l’art : cas d’usage élargis & rôle des laboratoires
Cas d’usage avancés pour l’art : de la suspicion à la preuve opposable
L’analyse post-bombe est particulièrement utile pour les professionnels du marché de l’art dans les cas suivants :
- Authentification d’œuvres modernes : elle peut prouver par exemple qu’un matériau date bien des années 1960–1970.
- Détection de faux ou d’imitations : elle permet d’identifier un matériau trop récent pour être en cohérence avec l’attribution annoncée.
- Conformité réglementaire (CITES) : elle différencie un ivoire pré-1975 (légal) d’un prélèvement plus récent (interdit).
- Contentieux ou assurance : elle est utilisée pour établir une preuve scientifique opposable en cas de litige ou de sinistre.
A ce titre voici quelques cas d’usage appliqués de l’analyse post-bombe pour les professionnels et collectionneurs d’art :
- Peinture « 19e » sur toile : avec datation du lin. Si un bomb-pulse est détecté, alors la toile est postérieure à 1954. On peut ainsi envisager une toile de remplacement, une œuvre apocryphe, ou une copie.
- Dessin sur papier : l’absence ou la présence de signal post-bombe permet de valider ou disqualifier une datation alléguée. L’analyse est ainsi particulièrement utile dans le cadre des archives ou manuscrits contestés.
- Assemblages, collages, marouflages : les datations croisées (support, doublage, colle) permettent de reconstituer la chronologie des interventions. Cette analyse est essentielle pour les dossiers d’assurance.
Réaliser une analyse post-bombe permet finalement de transformer un doute (incohérence stylistique, provenance lacunaire, signatures litigieuses) en élément scientifique objectivé, compréhensible par un comité, un tribunal ou une maison de ventes.
CIRAM : de l’échantillon à l’argumentaire d’authentification
Les équipes de scientifiques des laboratoires CIRAM interviennent de bout en bout pour accompagner les professionnels du marché de l’art. Leur action couvre plusieurs étapes :
- Cadrage : choix de la matière la plus probante (lin vs colle, bois du châssis vs clé, dentine vs ciment dentaire, etc…), proposition d’une stratégie de micro-prélèvement, anticipation des risques de contamination.
- Mesure AMS : mise en place de procédures dédiées aux matériaux organiques de l’art, référentiels NH/SH, corrections isotopiques, gestion des matrices mixtes.
- Interprétation & rédaction : création d’un rapport clair, citable, présentant les résultats (pMC/Fcarbone 14), intervalle calendaire post-bombe, incertitude, scénarios d’interprétation et conclusion motivée.
- Confidentialité : signature d’un NDA, anonymisation des échantillons, circuits sécurisés, chaîne de possession tracée. Cet aspect est un point central pour des dossiers sensibles.
Si la situation l’exige (en fonction de l’objet de l’étude), CIRAM réalise également des analyses complémentaires (FTIR pour liants et colles, dendrochronologie pour bois, imagerie pour interventions, pigments et/ou liants sur une couche picturale) afin de fournir une lecture matérielle cohérente.
Loin d’être une curiosité scientifique, l’effet post-bombe est devenu un outil incontournable pour l’authentification des œuvres d’art. Grâce à la datation carbone 14 post-1954, il est désormais possible d’apporter une preuve scientifique rigoureuse sur l’origine temporelle de matériaux organiques : bois, ivoire, lin, papier… Bien exploitée, l’analyse post-bombe renforce alors l’authentification, identifie les restaurations, sécurise les ventes et protège collectionneurs comme institutions. La clé d’une étude réussie réside dans des prélèvements adaptés, des protocoles AMS rigoureux et une interprétation contextualisée.
Avec son expérience des enjeux du marché de l’art, CIRAM transforme la mesure carbone 14 en preuve lisible, intégrable aux expertises, catalogues et procédures de due diligence. En sa qualité de laboratoire expert en datation et en caractérisation des matériaux pour le marché de l’art, CIRAM met à votre disposition ses compétences, ses équipements de pointe et son accompagnement sur mesure pour transformer chaque analyse carbone 14 en preuve exploitable, documentée et opposable.
Si vous souhaitez faire analyser votre ou vos objets d’art par le leader de la datation carbone 14 en France, contactez les équipes de CIRAM et demandez votre étude.