Carbon-14 dating is the most widely used dating method. It applies to all organic materials such as wood, charcoal, bone, teeth, paper, textiles, ivory and plant macroremains.
Find out more about its scope of action and the methods used to meet your needs.
The importance of carbon-14 in materials analysis
Radiocarbon analysis was developed in the 1940s and is based on the physical properties of the 14 isotope of carbon. This isotope is radioactive, meaning that it is unstable over time. It will disappear and be transformed into nitrogen. Every 5,730 years, the concentration of carbon 14 is halved, disappearing completely after 60,000 years. The quantity of carbon is stable for living beings, because on the one hand, carbon 14 disintegrates, but on the other, it is reintegrated through respiration or photosynthesis. This is why C14 dating will date the death of the individual (or plant), i.e. the moment when the disappearance/integration equilibrium is broken.
Cutting-edge technologies for carbon-14 analysis
For radiocarbon measurement, we use state-of-the-art technologies such as elemental analysis (EA), isotope ratio mass spectrometry (IRMS), automatic graphitization and particle gas pedal mass spectrometry (AMS). The AMS method requires very little material (1 mg of carbon vs. 1g previously), for a more accurate result than the liquid scintillation technique.
Essential analysis for archaeological digs
Charcoal and bone are very good chronological markers in an archaeological dig, as they are closely linked to the stratigraphy in which they are found. This is why carbon-14 dating is the most widely used scientific technique to support archaeological excavations. Depending on the period, dating accuracy can range from forty years to several centuries.
Authenticating works of art with radiocarbon analysis
Carbon 14 will also be used to authenticate works of art, such as wooden or ivory sculptures, or painting supports, such as textiles and paper.
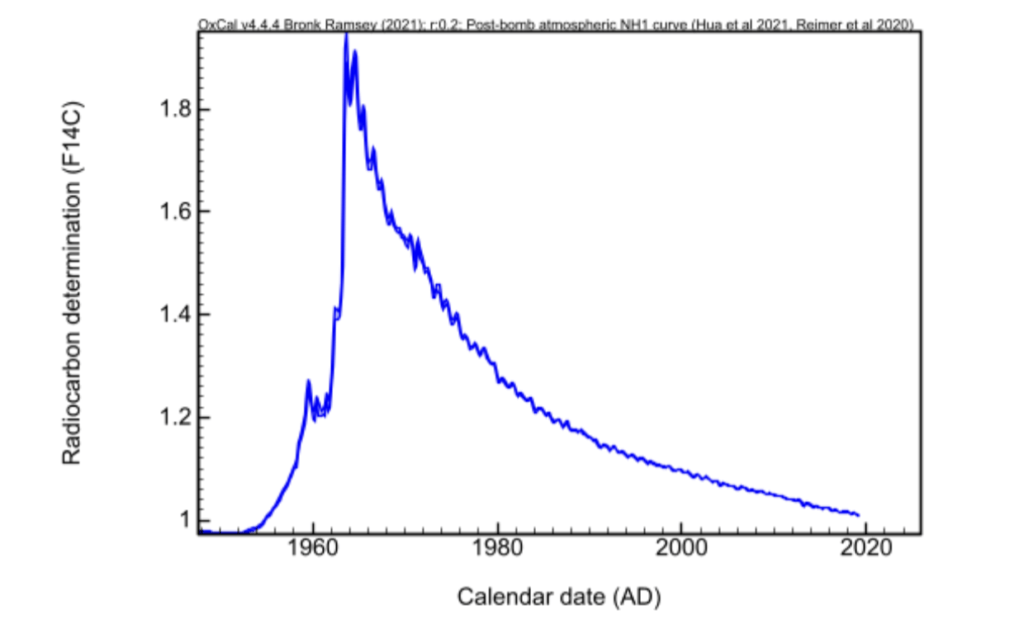
It's important to note that organisms that lived after 1950 have a very high 14C concentration, far exceeding the "classic" concentration of the carbon isotope 14 compared to the other two isotopes. This is mainly due to atmospheric nuclear testing, which resulted in massive radiocarbon production. That's why there's a real dating frontier in 1950. This boundary is ideal for formally identifying recent fakes. Moreover, post-1950 dating is exceptionally accurate.
C14 analysis in the food and industrial sectors
Radiocarbon analysis is also used in industry to qualify biobased products and quantify their biogenic and petroleum-based components. As mentioned above, after 60,000 years, carbon 14 has completely disappeared. As petroleum is derived from the decomposition of organic matter over millions of years, it no longer contains any 14C. Conversely, biomass (i.e. living organisms) is "full" of carbon-14. Radiocarbon analysis enables us to distinguish between the proportion of biosourced/biogenic carbon (modern) and fossil carbon (ancient) present in all types of material, whether solid, liquid or gaseous. We measure the ratio of 14C, 13C and 12C isotopes in a sample. Although the proportion of 14C is very low (1 atom of 14C for 1012 atoms of 12C), it is nevertheless present in all molecules containing modern carbon".